Enteric Coating Systems
Advanced acid-resistant coating systems engineered for targeted intestinal release and protection of acid-sensitive active ingredients.
Care Pharma Chem’s Enteric Coating Systems are designed to provide effective gastric resistance and targeted intestinal release for acid-sensitive active ingredients. These systems are formulated using high-performance methacrylic acid copolymers to ensure consistent dissolution behavior and regulatory compliance.
Available in both solvent-based and aqueous systems, our enteric coatings deliver reliable film integrity, controlled weight gain and optimized processing performance across standard coating equipment. We prioritize the stability of the active pharmaceutical ingredient (API) by ensuring the coating remains intact at gastric pH while providing rapid release at the desired intestinal site.
High-performance enteric systems for gastric protection and targeted delivery.
| Grade | Polymer Base | Reconstitution Level | Recommended Weight Gain |
|---|---|---|---|
| Cotab EN (Solvent-Based) | Methacrylic Acid Copolymer Type A | 8–9% in organic solvent (IPA 80% + MDC 20%) or 97% IPA + 3% Water or 70% IPA + 30% Acetone | 7.0 – 8.0% |
| Cotab E AQ (Aqueous System) | Methacrylic Acid Copolymer Type C | 15 – 20% (Aqueous) | 8.0 – 10.0% |
Provides effective gastric protection for acid-sensitive APIs.
Designed for rapid dissolution at higher pH levels (pH > 5.5).
Maintains coating strength and flexibility during processing.
Suitable for conventional and automated coating systems.
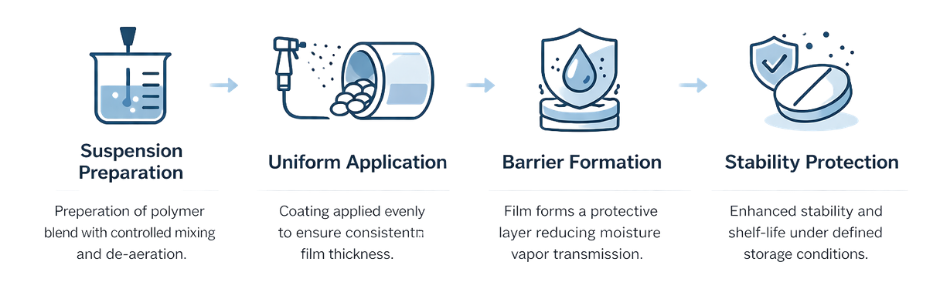
Homogeneous dispersion of methacrylic copolymers.
Precision spraying with monitored parameters.
Performance testing according to pharmacopeia.
Enteric systems are engineered to remain intact under acidic gastric conditions (pH 1.2) while dissolving at defined intestinal pH levels. Performance validation should be conducted according to pharmacopeial guidelines to ensure reproducibility and compliance.
