Application Guide
Technical guidance for suspension preparation, process optimization and controlled film application across pharmaceutical coating systems.

Effective pharmaceutical coating requires precise alignment between formulation properties and process parameters. This application guide outlines recommended preparation, dispersion control and process optimization steps to ensure consistent film formation and reproducible coating performance.
Care Pharma Chem systems are developed to support compatibility with conventional pan and fluid bed coating equipment under validated operating conditions. Our technical framework is designed to assist manufacturers in achieving high-quality coating finishes while maintaining process efficiency and regulatory compliance.
Technical Notes
Align spray rate with drying capacity to prevent overwetting and sticking.
Ensure fine droplet distribution for uniform film formation and smooth finish.
Support solvent evaporation without inducing film stress or core degradation.
Monitor process stability and drying efficiency throughout the coating cycle.

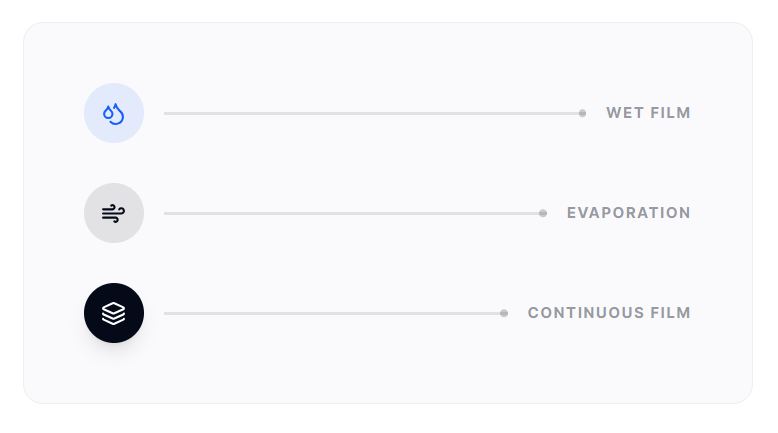
Film formation occurs as solvent evaporation enables polymer coalescence and consolidation. Controlled drying ensures smooth surface finish, mechanical integrity and adhesion without defects such as blistering or cracking.
Balance between spray rate and drying capacity is critical to achieving reproducible coating quality. Inadequate drying can lead to “picking” and “twinning,” while excessive drying can cause “spray drying” and rough surface textures.
Coating weight gain should be monitored at defined intervals to ensure uniform film thickness and performance consistency. Incremental sampling and in-process verification are recommended to prevent over-application and ensure regulatory compliance.